 Dead
pearl oysters due to Heterocapsa red tide(Ago Bay, 1992)
Dead
pearl oysters due to Heterocapsa red tide(Ago Bay, 1992)
When red tide occurrs, various marine
animals are affected by the red tide organism. In last three decades, Chattonella,
Gymnodinium
and Heterosigma were representative nuisance red tide genera off
the Japanese coast and these organisms have frequently caused serious damages
mainly to cultured finfish (Honjo 1994). These harmful genera damage the
epithelial tissues of the fish gills, subsequently depress the gas exchange
rate at gill surface. In H. circularisquama red tide, however, any
negative responses and dying of wild and farmed finfish has never recorded.
In some cases, caged fishes had normally swimming in the assemblages of
H.
circularisquama cells (>107 cells/L).
H. circularisquama red tide kills mainly bivalve
molluscs; pearl oyster Pinctada fucata, manila clam Ruditapes
philippinarum, pacific oyster Crassostrea gigas, blue mussel
Mytilus
galloprovincialis etc (see below).
 Dead
pearl oysters due to Heterocapsa red tide(Ago Bay, 1992)
Dead
pearl oysters due to Heterocapsa red tide(Ago Bay, 1992)
 Dead pacific oysters due to Heterocapsa red tide(Hiroshima Bay,
1995)
Dead pacific oysters due to Heterocapsa red tide(Hiroshima Bay,
1995)
 Dead blue mussel due to Heterocapsa red tide
(Hiroshima Bay, 1995)
Dead blue mussel due to Heterocapsa red tide
(Hiroshima Bay, 1995)
 Farming ground of manila clam destroyed by red tide(Hiroshima Bay, 1998)
Farming ground of manila clam destroyed by red tide(Hiroshima Bay, 1998)
 Blue crab and grass puffer in red tide area (Hiroshima Bay, 1998)
Blue crab and grass puffer in red tide area (Hiroshima Bay, 1998)
Further, several commercially important
gastropods: abalones Hariotis discus, top shell Turbo also
affected by the H. circularisquama red tide. However, other marine
animals i.e. crustacean, vertebrates are not affected by the H. circularisquama
red tide. As the results, harmful dinoflagellate which kills only bivalve
and gastropod has not been reported.
2) Effects of Heterocapsa circularisquama on bivalve physiology
Laboratory experiment (Nagai et al. 1996) demonstrated that pearl oysters spat exposed to >106 H. circularisquama cells/L contracted their mantles markedly, closed their valves, and paralyzed, in some cases, the heartbeat of the juveniles completely ceased, and then they died. These negative symptoms were closely related to those of previous field observations (Matsuyama et al. 1996). Mortality increased markedly with the cell density of up to 2×107 H. circularisquama cells/L. Heart beat arrest or termination were occurred in both juvenile and adult pearl oysters above 106 H. circularisquama cells/L exposure within 30 minutes (Kiyohito NAGAI, per. comm.). In addition to lethal effect, the spat of pearl oyster significantly reduced their filtration rate when exposed to 5×104 H. circularisquama cells/L, and markedly decreased with increasing H. circularisquama cell densities (Matsuyama et al. 1997). This findings implicated that bivalves considerably ceased filtering activities at only one percent of red tide cell density.
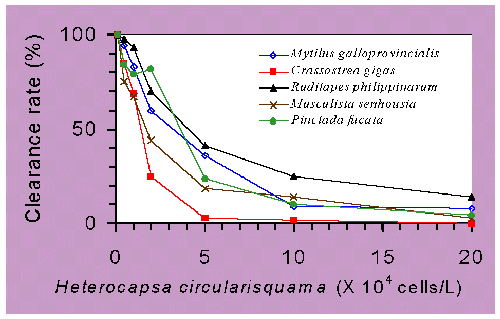 Relationships between H. circularisquama cell densities and clearance
rates of the several bivalve molluscs
Relationships between H. circularisquama cell densities and clearance
rates of the several bivalve molluscs
3) Toxic nature of Heterocapsa circularisquama
In the past, harmful red tide organisms
have frequently caused serious damages mainly to cultured finfish, with
bivalves to be a minor groups of damaged animals. Previous mortalities
of bivalves have been recorded only after the red tides caused by oxygen
depletion in association with sulfide production as a result of disintegration
of red tides. In the previous surveys, major death of bivalve due to H.
circularisquama was observed early to middle period of the red tide
outbreaks when dissolved oxygen was still near saturated (Matsuyama et
al. 1996). It was also observed that mortality of wild and cultured fish
was not the case during the red tide. From these facts, H. circularisquama
has a toxicity specific for bivalves molluscs.
According to previous study, the mussel
M.
galloprovincialis significantly reduced filtration rate at 5×104H.
circularisquama cells/L presence, accompanied with contraction of mantle
edges and exhalant siphons (Matsuyama et al. 1997). However, no inhibitory
effects on the filtration rate of mussels were observed with the filtrate
of a dense H. circularisquama culture (108 cells/L).
Therefore, it seems likely that H. circularisquama cells do not
produce extracellular metabolites which repress bivalve clearance rates
apart from in their cell walls. Furthermore, some physical treatment on
intact cell of H. circularisquama markedly inactivated the toxicity
on filtration rate. Supplemental experiment demonstrated that toxic nature
of H. circularisquama localized in cell surface (Matsuyama et al.
1997). The results of some treatment on
H. circularisquama cells
by surfacant and enzyme indicated that labile protein-like agent is probably
a toxic nature of H. circularisquama (Matsuyama et al. 1997) which
causes sustained disturbance of homeostasis and consequent death of shellfish
life.
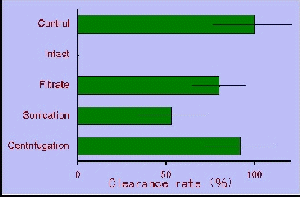 Effect of physical treatment on the toxicity of H. circularisquama
Effect of physical treatment on the toxicity of H. circularisquama
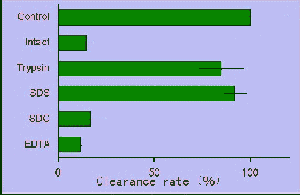 Effect of chemical treatment on the toxicity of H. circularisquama
Effect of chemical treatment on the toxicity of H. circularisquama
Otherwise, there is a possible concern
to the human illness responsible for the consumption of bivalve which accumulated
the
H. circularisquama cells. However, No human poisoning and shellfish
poisoning have been reported during the bloom of H. circularisquama.
Direct HPLC analysis showed that neither PSP toxins nor DSP toxins were
detected in the cells of H. circularisquama (S. Sakamoto and T.
Suzuki, pers. comm.). Mouse bioassay on the oysters collected from red
tide area showed no toxicity. On the basis of previous studies obtained
until now, it is concluded that there are no affair of human illness involving
with H. circularisquama bloom. Threfore, toxic mechanism of H.
circularisquama is greatly different from those of shellfish poisoning
causing genella
Alexandrium,
Dinophysis and Pseudo-nitzschia
spp..
4) Possible ecological meaning of toxicity on animals
In general, some phytoplankton produce
"phycotoxins" to prevent from predation and exclude the competitor in the
ecosystems. Protozoa, zooplankton, nekton (jellyfish), and filter-feeders
(bivalve, ascidian) are regarded as the major active predator on dinoflagellate
H.
circularisquama. Recent studies revealed that H. circularisquama
inhibit not only filter-feeder physiology but also their active predator
and competitor such as protozoa (Kamiyama and Arima 1997) and mixotrophic
dinoflagellate (Uchida et al. 1995). Thus, the inhibitory effect of H.
circularisquama upon their active predator and competitor might, itself,
be of great advantage for the development of red tide